Introduction
Traditionally, herbal medicine has been used to treat or prevent diseases. Galgeun-tang (GGT, gegen-tang in China and kakkon-to in Japan), one of the more common herbal formulas, includes seven herbs: Puerariae Radix (major component), Cinnamomi Cortex Spissus, Ephedra Herba, Paeoniae Radix, Glycyrrhizae Radix et Rhizoma, Zingiberis Rhizoma Crudus and Zizyphi Fructus. GGT is used to treat the common cold, fevers, headaches, hangovers and neck and upper back stiffness. Recently, several studies have shown that GGT is effective in alleviating influenza infection1,2) and allergic diarrhea3) and in immunomodulation.4) However, the efficacy of GGT in treating AD has not yet been examined.
Atopic dermatitis (AD) is an inflammatory, chronically relapsing, non-contagious and pruritic skin disorder.5) AD is often accompanied by allergic inflammation, which is initiated by activation of the adoptive immune response. Immunoglobulin E (IgE) is produced in plasma cells and bound by mast cells in type I allergic reactions. The IgE-primed mast cells release chemical mediators such as histamine, leukotrienes (LTs) and prostaglandin D2 (PGD2). These mediators lead to immediate phase reactions in the tissue, such as redness and itching, shortly after allergen-IgE binding. In the later phases of the disease, cytokines (IL-4 and IL-13) and chemokines are generated and released several hours after allergen-antibody cross-linking.6)
Topical corticosteroids are currently the most potent treatment for AD. However, patients with more severe forms of the disease do not always respond satisfactorily to these agents. Chronic use can also be associated with significant adverse effects. Because long-term application of corticosteroids results in tachyphylaxis and treatment resistance, it would be advantageous to develop new treatments that lack the side effects of corticosteroids.7) Systemic corticosteroids are known to be effective in the short-term treatment of AD, but no studies exist to support their long-term use, and rebound flaring and long-term side effects are limiting.5) Immuno-suppressive drugs, including calcineurin inhibitors such as cyclosporine, tacrolimus and pimecrolimus have been reported to be effective for atopic dermatitis, but concerns over systemic toxicity have limited their use.9, 10) Tacrolimus has been developed for the treatment of moderate to severe AD, but topical tacrolimus ointment causes transient burning in ∼60% of patients.10) Consequently, the need to efficiently manage the AD response while reducing side effects has led to the development of alternative remedies.
In the present study, we evaluate the anti-inflammatory and anti-allergic effects of GGT by measuring its inhibition of nitric oxide (NO) and PGE2 production in lipopolysaccharide (LPS)-treated RAW264.7 cells, phorbol-12 myristate 13-acetate (PMA)/A23187-stimulated histamine production in MC/9 cells, thymus and activation-regulated chemokine (TARC/CCL17) expression in TNF-α/IFN-γ-treated HaCaT cells and the AD response in NC/Nga mice.
Materials and Methods
1. Preparation of Galgeun-tang (GGT) extract
The seven constituents of GGT (Table I) were chopped, mixed and extracted in water at 100°C for 120 min in an herb extractor (COSMOS660, KyungSeo Machine Co., Incheon, Korea) and then filtered. All GGT components were taxonomically confirmed by Professor Je-Hyeun Lee, Dongguk University, Korea. A voucher specimen (2008-KE02-1 to 2008-KE02-7) has been deposited at the Herbal Medicine EBM Research Center, Korea Institute of Oriental Medicine. The extract was lyophilized (yield; 12.6%). Powdered GGT extract was stored at 4°C.
2. Cell culture and viability assay
RAW 264.7 murine macrophage cells and MC/9 murine mast cells were obtained from the American Type Culture Collection (ATCC, Rockville, MD, U.S.A.). The RAW 264.7 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM, Gibco BRL., NY, U.S.A.) supplemented with 5.5% (v/v) fetal bovine serum (FBS, Gibco BRL., NY, U.S.A.), 100 U/mL penicillin and 100 μg/mL streptomycin (Gibco BRL., NY, U.S.A.). The MC/9 cells were maintained in DMEM media containing 10% (v/v) FBS, 0.05 mM 2-mercaptoethanol (Sigma Chemical Co., MO, U.S.A.), 10% (v/v) Rat T-STIM (BD Biosciences, MA, U.S.A.), 100 U/mL of penicillin and 100 μg/mL of streptomycin in a humidified 5% CO2 atmosphere. HaCaT cells, human keratinocytes provided by Professor Na Gyong Lee (Sejong University, Korea), were cultured in DMEM with 10% (v/v) FBS and 100 U/mL of penicillin and 100 μg/mL of streptomycin.
RAW 264.7, MC/9 and HaCaT cells were seeded at densities of 2.5×103, 2.5×103 and 1.0×103 cells/well, respectively, in 96-well plates. The cells were then incubated with different concentrations of GGT extract (7.8–500 μg/mL) for 24 hr, with 1% DMSO serving as the control. After treatment, 10 μL of Cell Counting Kit-8 reagent (CCK-8, Dojindo, Japan) was added to each well, and the plates were incubated for 4 hr. Absorbance was measured at 450 nm using a microplate reader (BenchmarkPlus, Bio-Rad Laboratories Inc., U.S.A.) and the percentages of viable cells were calculated. Non-cytotoxic concentrations of GGT were used for all subsequent experiments.
3. Measurement of nitric oxide (NO) and PGE2 production in LPS-treated RAW264.7 cells
RAW 264.7 cells were seeded at a density of 2.5×105 cells in a 48-well plate for the NO and PGE2 assay. After resting for 18 hr, the cells were stimulated with 1 g/ml of LPS in the presence or absence of GGT extracts (2, 20 and 200 μg/mL) for 18 hr. NG-methyl-L-arginine (NMMA; Sigma-Aldrich, Inc., MO, U.S.A.) and indomethacin (Sigma-Aldrich, Inc., MO, U.S.A.) were used as positive controls. A Griess reagent system (Promega, WI, U.S.A.) was used to measure the production of NO in culture supernatants. Briefly, samples (50 μL/well) were incubated at room temperature with 1% sulfanilamide for 10 min and 1% α-naphthylamine for 10 min. The absorbance was then evaluated at 535 nm using a calibration curve with standard.
4. Measurement of histamine in PMA/A23187-treated MC/9 cells
MC/9 cells were plated in 48 well plates with 2×105 cells. The cells were either untreated or treated with phorbol 12-myristate 13-acetate (50 nM; PMA, Sigma-Aldrich, Inc., MO, U.S.A.)/A23187 (1 μM; Sigma-Aldrich, Inc., MO, U.S.A.) or PMA/A23187 (PA) + GGT (50–200 μg/mL) for 24 hr. Histamine levels in the MC/9 cell supernatants were measured by ELISA according to the manufacturer’s instructions (Oxford Biomedical Research, U.S.A.).
5. Measurement of TARC in TNF-α/IFN-γ-treated HaCaT cells
HaCaT cells were seeded at 1×106 cells/well in 6-well plates until they reached confluence. The cells were washed with serum-free media and incubated with 1 mL of serum-free media containing TNF-α (10 ng/mL), IFN-γ (10 ng/mL) and GGT extract (0, 125, 250 or 500 μg/mL) for 24 hr. Silymarin (12.5, 25 or 50 μg/mL) was used as a positive control. Human TARC levels in the culture supernatants were measured by ELISA according to the manufacturer’s instructions (R&D Systems, U.S.A.).
6. Animals and sensitization
Male NC/Nga mice (8 weeks old) were obtained from Central Laboratory Animal Inc. (Seoul, Korea), housed individually in an air-conditioned room and maintained at 24±2°C with 55±15% relative humidity. The animals were allowed to acclimatize for 2 weeks before the experiments were initiated. All procedures involving animals were conducted in accordance with the Guideline of the institutional Animal Care and Use Committee of the Korea Institute of Oriental Medicine. The approval number for the animal study was #10-052.
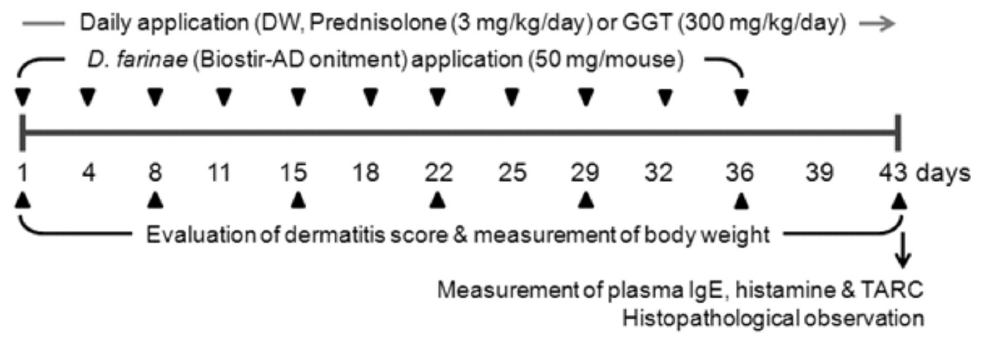
AD-like skin lesions were induced in male NC/Nga mice using Dermatophagoides farina allergen extract ointment (Biostir-AD, Biostir Co., Ltd., Kobe, Japan) (Hirotoshi et al., 2004). The mice (10 weeks old) were grouped randomly into four groups with seven mice per group. The mice were divided into untreated (normal; distilled water, p.o.), Biostir-AD-treated (control; distilled water, p.o.), Biostir-AD plus prednisolone-treated (Prednisolone; 3 mg/kg/day, p.o.) and Biostir-AD plus GGT extract-treated (GGT; 300 mg/kg/day, p.o.) groups. For sensitization, 50 mg Biostir-AD was applied topically on the upper dorsal skin and the back of the ears of each mouse twice weekly for 6 weeks. Prednisolone, the positive control, and GGT were each dissolved in distilled water and administrated daily to mice for 6 weeks (Fig. 1).
7. Dermatitis score
The dermatitis scores were assessed by evaluating the dorsal skin and the ears of the mice once a week for 6 weeks. The Eczema Area and Severity Index (EASI) scoring system was employed to evaluate the severity of dermatitis. The dermatitis score was defined as the sum of the scores for erythema/hemorrhage, edema, excoriation/erosion and scaling/dryness and scored as follows: no symptoms, 0; mild, 1; moderate, 2; and severe, 3.8)
8. Histological analysis
Mice were anaesthetized using pentobarbital sodium (Entobar inj., Hanlim Pharm. Co., Ltd., Korea) injection (i.p.). Blood samples were taken, and the animals were sacrificed by exsanguination from the aorta. Complete gross observation was performed on all terminated animals. The blood samples were collected into a microtainer (Becton, Dickinson and Company, NJ, USA) containing K2-EDTA. Plasma samples were collected after centrifugation at 10,000 rpm and stored at −80°C until they were further assayed. The dorsal skin and one ear of each mouse were removed and fixed in 10% (v/v) natural buffered formalin for 24 hr. Tissues were embedded in paraffin, sectioned at 4 m thickness, and stained with hematoxylin & eosin (H&E) or toluidine blue to estimate epidermal inflammation (hypertrophy and infiltration by inflammatory cells) and mast cell counts, respectively. Dermal mast cells were counted as the number of toluidine-blue positive cells in randomly selected high power fields on each specimen using a light microscope.
9. Plasma levels of IgE, histamine and mTARC
Plasma levels of IgE (Bethyl Laboratories Inc., U.S.A.), histamine (Oxford Biomedical Research, U.S.A.) and mTARC (R&D Systems, U.S.A.) were measured by ELISA, according to the manufacturer’s instructions.
10. Statistical analysis
Data were expressed as meanSEM and analyzed using one-way ANOVA followed by the Bonferroni multiple comparison method. A P-value <0.05 was defined as statistically significant. All statistical analyses were performed using the SYSTAT® 8.0 program (SYSTAT Inc., Evanston, IL, U.S.A.).
Results
1. Inhibition of NO, PGE2, histamine and TARC production by GGT
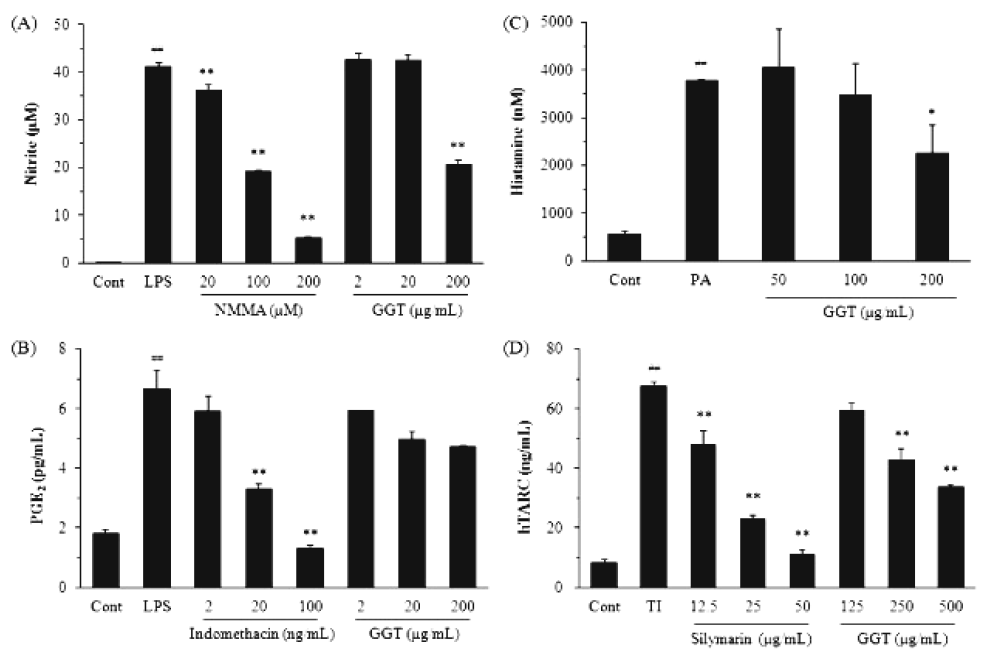
GGT was shown to inhibit the inflammatory response in LPS-stimulated RAW264.7 cells. LPS-induced NO production was significantly reduced by 49.7±1.9% following treatment with 200 μg/mL GGT extract compared to the group treated with LPS alone (P<0.01, Fig. 2A). PGE2 production, however, was not significantly altered (Fig. 2B). PA-stimulated MC/9 cells treated with GGT showed a slight decline in histamine production compared to those stimulated with PA alone (P<0.05, Fig. 2C). GGT inhibited TARC production by TNF-α/IFN-γ-stimulated HaCaT cells in a dose-dependent manner (P<0.01, Fig. 2D).
2. Dermatitis scores in Nc/Nga mice
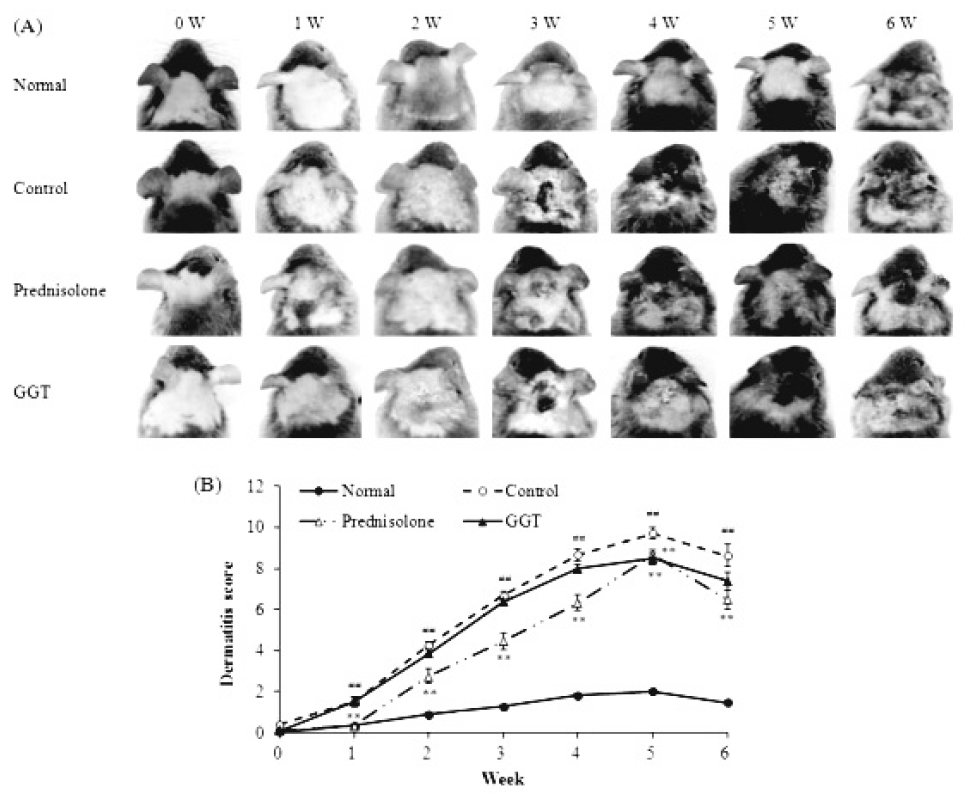
Typical photographs of each group during the experiment are shown in Fig. 3A. Macroscopically, the mice developed lesions on the dorsal skin and ears starting at 1 week after the initiation of Biostir-AD application. In the prednisolone-treated positive control group, the dorsal skin and ear severity was significantly reduced compared to the control group (P<0.01). However, the dermatitis score of the GGT-treated group was no different than that of the control group with exception of the fifth week of treatment (P<0.01, Fig. 3B). The maximum dermatitis score was recorded following the fifth week of Biostir-AD application in all groups, and the scores for each group were as follows: 2.0±0.07 (normal), 9.7±0.25 (control), 8.7±0.22 (prednisolone) and 8.5±0.26 (GGT) (Fig. 3B).
3. Histological observations
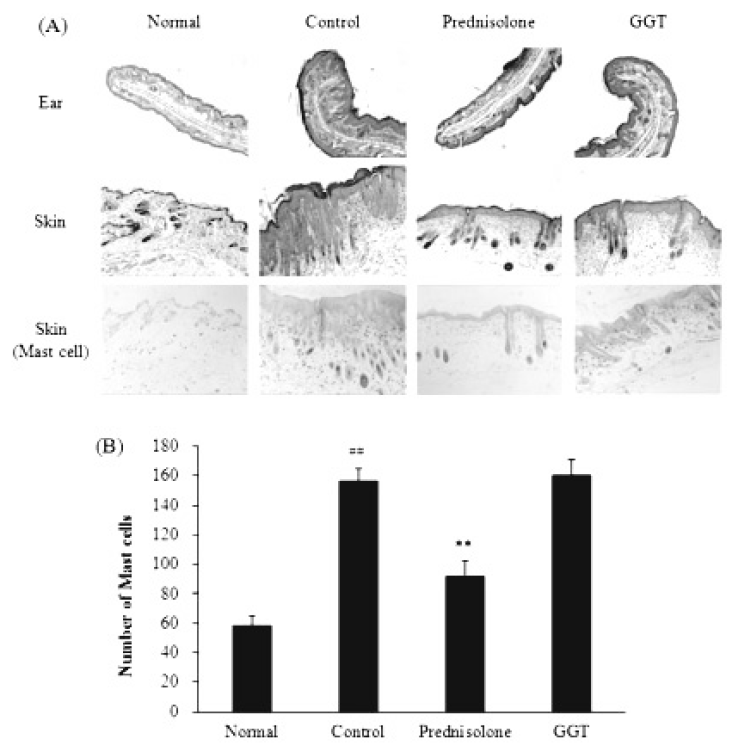
There were no histological differences observed in the dorsal skin or ear lesions between the GGT-treated and untreated mice. Mast cell infiltration in the dorsal skin was not reduced by administration of GGT to AD-induced mice (Fig. 4).
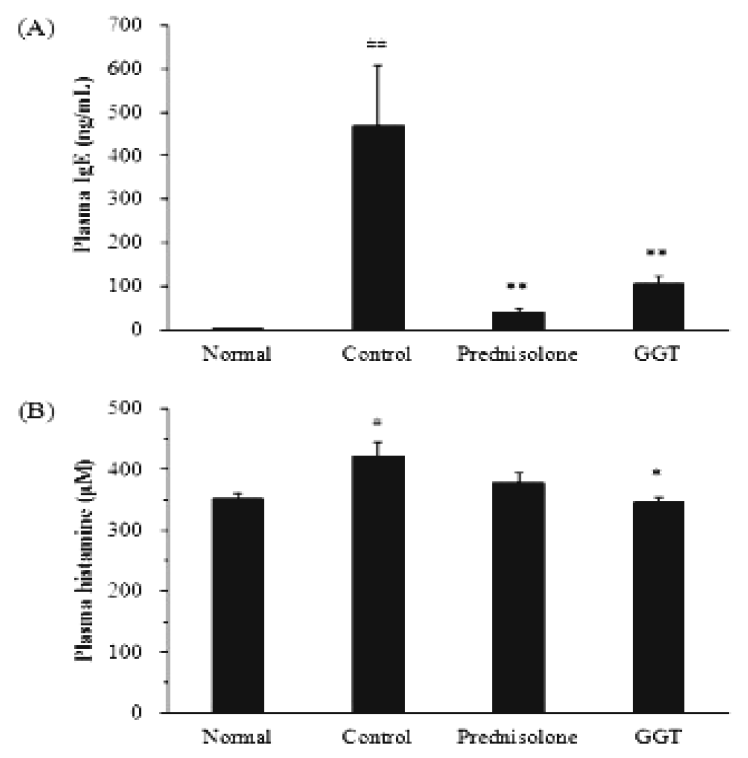
4. Plasma levels of IgE, histamine and mTARC
Total plasma IgE levels were significantly increased in the control group (469.9±138.3 ng/mL) compared with those of the normal group (2.4±0.9 ng/mL, P<0.01). Prednisolone (39.7±9.7 ng/mL) and GGT (105.8±16.9 ng/mL) inhibited increases in plasma IgE levels in Biostir-AD-sensitized mice (P<0.01, Fig. 5A). The plasma histamine levels in the control group (421.2±23.6 M) were elevated compared to the normal group (352.6±9.0 μM, P<0.05). The histamine levels were reduced in the GGT-treated mice (346.1±9.2 μM) compared to the control group (P<0.05); however, there was no significant difference compared to the prednisolone-treated group (377.2±17.4 μM, Fig. 5B). GGT did not reduce mTARC levels following Biostir-AD application in NC/Nga mice (data not shown).
Discussion
Atopic dermatitis (AD) is a common chronic cutaneous disease characterized by chronic skin inflammation, cutaneous erythema, induration, severe pruritus, over-expression of IL-10 and high IgE levels. The most important allergens associated with human AD are house dust mite allergens, and Dermatophagoides farina is the most common house dust mite present in the environment. Inflammation in AD is biphasic. An initial Th2 phase leads to a chronic phase associated with Th0 and Th1 cells.5) The drugs currently used to treat AD are limited by the significant adverse effects associated with their long-term usage. Recently, several studies have attempted to find new candidates to treat AD with fewer side effects. Herbal remedies, including traditional herbal formulas are a popular trend in the field of complementary and alternative medicine. The worldwide public is using increasing numbers of herbal remedies in conjunction with or as replacements for conventional medicine.11) However, physicians, pharmacists and consumers are besieged with questions about the safety and efficacy of herbal remedies.12)
GGT, an herbal formula, has been traditionally used to treat infectious diseases in East Asia. In our previous study, the chemical contents of the GGT extract were analyzed using a high performance liquid chromatography (HPLC) system, and we measured the presence of albiflorin, paeoniflorin, cinnamaldehyde, cinnamic acid, glycyrrhizin, liquiritin, daidzin and puerarin.13) These ingredients have been reported to inhibit inflammatory responses.14–17) Because AD is strongly associated with the inflammatory response, we investigated the anti-inflammatory and anti-AD effects of GGT treatment using in vitro and in vivo systems.
Treating LPS-stimulated RAW 264.7 cells with GGT reduced NO production. Keratinocyte exposure to TNF-α and IFN-γ has been shown to induce the expression of cytokines and chemokines that mediate the infiltration of monocytes and T cells to sites of inflammation in the skin. TNF-α and IFN-γ synergistically induces TARC production in human keratinocytes and HaCaT cells.18) C-C chemokine receptor 4 (CCR4) was found to be predominantly expressed by Th2 cells, and high macrophage-derived chemokine (MDC/CCL22) production has been observed in AD patients and, in association with TARC/CCL17, in NC/Nga mice exhibiting atopic dermatitis-like lesions. The involvement of Th2 cells both helps to explain the joint involvement of IgE-producing B cells (via IL-4 and IL-13), mast cells (via IL-4 and IL-10) and eosinophils (via IL-5) in the allergic inflammatory process and accounts for the other pathophysiologic features of allergy.19) Th2-associated chemokines (TARC and MDC) are elevated in the serum and in epidermal keratinocytes of AD patients compared with normal control subjects. These levels correlate with disease severity, serum IgE levels, and blood eosinophil numbers. 20,21) GGT suppressed both the production of histamine in PMA/A23187-stimulated MC/9 mast cells and the expression of TARC in TNF-α/IFN-γ-treated HaCaT keratinocytes.
NC/Nga mice, which are characterized by AD-like skin lesions, show elevated levels of blood IgE.22) Mast cell infiltration is another important factor in AD development. Dermatitis scores were reduced following GGT treatment after the fifth week in house dust mite-sensitized NC/Nga mice. However, GGT did not suppress the histological features of the disease, including edema, cornification of the epidermis, and mast cell infiltration in the dorsal skin and ear. GGT reduced the plasma levels of histamine and IgE in house dust mite extract-sensitized NC/Nga mice. A single dosage of GGT is approximately 44.0 g of dried herb, which is equivalent to 5.54 g of the extract (yield of extract = 12.6% of the raw material) in humans. Considering the average body weight of an adult as 60 kg, the normal dosage for a 60 kg human is 92.4 mg of GGT extract/kg. The dose equivalent per kg for mouse (12.3) was derived by dividing the Km factor, the body surface area (m2) to body weight (kg) ratio, for humans by the Km factor for mice,23) the result being a mouse dose equivalent of 1,136 mg/kg (92.4 mg/kg×12.3). In the present study, the dose of GGT extract used, 300 mg/kg in mice, was just more than a quarter of a single human dose.
Conclusions
We suggest that GGT reduces the atopic dermatitis response of dust mite extract-sensitized NC/Nga mice via inhibition of IgE-mediated mast cell degranulation and by showing the release of preformed mediators such as histamine. This leads us to conclude that GGT should be explored as a potential therapeutic agent for treating atopic dermatitis.