Introduction
Benign prostatic hyperplasia (BPH), an aging-related chronic disease in men, is a noncancerous increase in size of the prostate caused by hyperplasia of prostatic stromal and epithelial cells. Prostate hyperplasia results in lower urinary tract symptoms (LUTS), such as urinary frequency, urgency, nocturia, weak stream, hesitancy, intermittency, incomplete emptying and straining, because the enlarged prostate leads to urethral compression. Commonly used medications to treat BPH are classified as α-adrenergic receptor blockers, 5-α reductase inhibitors (5-ARIs), phosphodiesterase-5 (PDE-5) inhibitors, anticholinergic and β-3 agonists. The limitations of these medications are weak or moderate efficacy, long-term administration and side effects such as lowering of blood pressure, dizziness, and peripheral edema1). Recently, it has been cited that not only do the combination of α-blocker/5-ARI, PDE-5 inhibitor/α-blocker, and PDE-5 inhibitor/5-ARI have additive effects compared to monotherapy in LUTS-BPH patients2), but also several herbal remedies, such as complementary and alternative medications, have a positive influence against a low degree of efficacy and short- and long-term adverse effects of conventional drugs against BPH3).
In recent years, the use of herbal remedies has been increasing as the awareness of complementary and alternative medicine (CAM) has raised due to the growing interest in health care and the therapeutic effects of conventional medicines that do not meet the expectations of patient4). Yongdamsagan-tang (YST; Long Dan Xie Gan-tang in Chinese and Ryutan-shakan-to in Japanese) and Paljung-san (PJS; Ba Zheng-san in Chinese), traditional herbal formulas, contain eleven and nine herbal medicines, respectively. YST and PJS are known to be used to treat BPH due to their ‘clear heat and drain dampness’ effects5). YST has been traditionally used to treat liver and/or gallbladder-related symptoms, such as headache, hypochromic pain, dizziness, and difficult urination with pain. Recently, it has been cited that YST has not only immunomodulatory, hepatoprotective, anti-inflammatory and anti-allergic effects in non-clinical studies6–8) but also reduction of postoperative pain and discomfort after transurethral resection of the prostate (TURP) and urination difficulty in clinical studies9,10). Traditionally, PJS has been used to treat urinary disturbances, including urinary retention, hematuria, dysuria, delayed micturition, and residual urine sense, in men. Recently, it has been reported that PJS reduces the international prostate symptom score (IPSS) and post void residual urine (PVR) in patients with BPH11), as well as infiltration of monocytes/macrophages and gene expression of inflammatory cytokines in the prostate of chronic nonbacterial prostatitis-induced rats12).
Chronic inflammation of the prostate is also known to be involved in the pathogenesis of BPH. In BPH patients, the prostate volume in the high-inflammation group was larger than in the low-inflammation group, and immune cell-mediated inflammation as well as stromal and epithelial cell-related inflammation were observed in the prostate13). T lymphocytes and macrophages are mainly related with inflammatory responses in the prostate with BPH14). In a previous study, our research group has reported that YST inhibits testosterone-induced benign prostatic hyperplasia through antiproliferative and antioxidative activities in the rat prostate15) and suppresses inflammatory response as well as cell proliferation in benign prostatic hyperplasia epithelial-1 (BPH-1) cells16). PJS has also been reported to induce apoptosis of BPH-1 cell and to inhibit prostate hyperplasia in rats17). Finasteride is a 5-ARI, which inhibits conversion from testosterone to dihydrotestosterone (DHT), which mediates prostate growth.
The combined use of herbal medicines and conventional medicines is increasing with the expectation of a higher effect18–19). However, no clinical studies on combination therapy of YST or PJS with finasteride have been found. In the present study, therefore, we evaluated both the anti-inflammatory activities of finasteride and two herbal formulas, YST and PJS, which have been used to treat BPH, on a macrophage cell line, RAW 264.7 cells, and the influence of herbal formulas on the activity of human drug metabolizing enzymes to predict herb-drug interactions when herbal formulas are used in combination with the conventional medication such as finasteride.
Materials & methods
1. Preparation of herbal formula extracts
The eleven and nine herbal constituents of YST and PJS (Table 1), respectively, were chopped, mixed and then extracted with 10-fold (w/v) water at 100°C for 2 h in an electric herb extractor (COSMOS-660, Kyungseo Machine Co., Incheon, Korea). All herbal constituents were identified by Professor Je Hyun Lee (Department of Herbology, College of Oriental Medicine, Dongguk University, Gyeongju, Korea) and Professor Young-Bae Seo (Department of Herbology, College of Oriental Medicine, Daejeon University, Daejeon, Korea). The voucher specimens (2012-KE49-1~11 and 2015-KE56-1~9) have been deposited at the K-herb Research Center, Korea Institute of Oriental Medicine. The extracted solutions of YST and PJS were filtered with a standard sieve (270 mesh) and lyophilized by using a freeze drier (PVT100, Ilshin Bio Base, Yangju, Korea). Yields of the powdered YST and PJS extract were 18.53% and 9.34%, respectively. The phytochemical ingredients of two herbal formulas were analyzed in previous reports16,17). The contents of phytochemical ingredients of both herbal formulas are described in Table 2.
2. Cell culture and viability assay
The murine macrophage cell line RAW 264.7 was obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA). The cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM, Gibco BRL, Grand Island, NY, USA) containing 5.5% (v/v) heat-inactivated fetal bovine serum (FBS, Gibco BRL), 100 U/ml penicillin (Gibco BRL) and 100 μg/mL streptomycin (Gibco BRL) at 37°C in a humidified, 5% CO2 atmosphere. To assay cell viability, cells (3×103 cells/well) were seeded in 96-well plate and incubated for 16 h. The cells were treated with various concentrations (0, 6.25–400 μg/mL) of herbal formula extracts. After treatment for 24 h, 10 μL of Cell Counting Kit-8 reagent (CCK-8, Dojindo Lab., Kumamoto, Japan) was added to each well. After incubation with CCK-8 for 4 h, the absorbance of each well was measured at 450 nm using a Benchmark Plus Microplate Reader (Bio-Rad Lab., Hercules, CA, USA) and the percentages of viable cells were calculated. Non-cytotoxic concentrations of extracts were used for subsequent assays.
3. Quantification of inflammatory mediators and cytokines in LPS-stimulated RAW 264.7 cells
RAW 264.7 cells were plated in 48-well plates at 2.5×105 cells/well. After resting for 16 h, the cells were stimulated with 1 μg/mL lipopolysaccharide (LPS; Sigma Chemical Co., St. Louis, MO, USA) in the presence or absence of herbal formula extracts and finasteride (Sigma Chemical Co.) for 24 h. The positive controls for nitric oxide (NO) and prostagladin E2 (PGE2) inhibition were NG-methyl-L-arginine (NMMA; Sigma Chemical Co.) and indomethacin (Sigma Chemical Co.), respectively. The cultured media were collected and the levels of nitrite (as an index of NO), PGE2, interleukin-6 (IL-6) and tumor necrosis factor (TNF-α) were quantified. The levels of inflammatory mediators (NO and PGE2) and cytokines (IL-6 and TNF-α) were measured by the Griess reagent system (Promega Co., Madison, WI, USA), PGE2 enzyme-linked immunoassay (EIA) kit (Cayman Chemical Co., Ann Arbor, MI, USA) and BD OptEIA™ mouse IL-6 and TNF-α ELISA kits (BD Bioscience, San Jose, CA, USA), respectively, according to the manufacturer’s protocols. All assays were repeated at least three times.
4. Measurement of human hepatic microsomal cytochrome P-450s (CYP450s) activities
Vivid® CYP450 Screening Kits (Invitrogen Co., Camarillo, CA, USA) were used to measure the activities of human hepatic CYP1A1 (Vivid® CYP1A2 Blue), CYP2B6 (Vivid® CYP2B6 Blue), CYP2C9 (Vivid®CYP2C9 Blue), CYP2C19 (Vivid® CYP2C19 Blue), CYP2D6 (Vivid® CYP2D6 Blue), CYP2E1 (Vivid® CYP2E1 Blue) and CYP3A4 (Vivid® CYP3A4 Blue) according to the manufacturer’s protocols. In brief, 40 μL of 2.5× test samples and each known CYP450 isozyme inhibitors as positive controls were added into the wells of a black 96-well plate for fluorescence (Thermo Fisher Scientific, Roskilde, Denmark). α-Naphthoflavone, sulfaphenazole, quinidine, sodium diethyldithiocarbamate trihydrate (DDC) and ketoconazole (Sigma Chemical Co.) were used as positive controls of enzyme activity inhibition for CYP1A2, CYP2C9, CYP2D6, CYP2E1 and CYP3A4, respectively. The positive control for CYP2B6 and CYP2C19 was miconazole (Sigma Chemical Co.). 50 μL of Master Pre-Mix solution containing CYP450 BACULOSOMES® Plus reagent as human microsomal P450 isozymes and Vivid® Regeneration System, including glucose-6-phosphate and glucose-6-phosphate dehydrogenase, in Vivid® CYP450 Reaction Buffer was added to each well. To encourage interaction of the test samples with CYP450 isozymes in the absence of enzyme turnover, the plate was incubated with shaking for 10 min at room temperature (RT). The enzyme reaction was started by adding 10 μL of 10× Vivid® Substrate and Vivid® NADP+ mixture. Vivid® EOMCC for CYP1A2, CYP2C19, CYP2D6 and CYP2E1 and Vivid® BOMCC for CYP2B6, CYP2C9 and CYP3A4 were used as a Vivid® Substrate. The plate was immediately transferred into a SpectraMax® i3 multi-mode plate reader (Molecular Devices Co., Sunnyvale, CA, USA), and fluorescence intensity (FI) was subsequently measured for 60 min at excitation (415 nm) and emission (460 nm) wavelengths. In the linear section of the fit to the FI and incubation time, the percent inhibition of CYP450s by test samples were calculated by the equation: % Inhibition = (1 – ΔT/ΔC) × 100%, where ΔT and ΔC are the difference in FI before and after incubation with the test samples and the control (no inhibitor), respectively. The data are described as the means ± standard error of the mean (SEM; n = 3). To calculate the 50% inhibitory concentration of the desired activity (IC50), the inhibition curve for enzyme activity by test samples was created using a SigmaPlot software Ver. 12.5 (Systat Software, Inc., CA, USA) capable of generating a 4-parameter logistic curve-fit.
5. Measurement of human microsomal UDP-glucuronosyltransferases (UGTs) activities
UGT1A1 and UGT2B7 activity were evaluated by using the UGT-Glo™ UGT1A1 and UGT2B7 Screening Systems (Promega, Madison, WI, USA), respectively, according to the manufacturer’s protocols. UGT1A4 activity was measured by using the recombinant human UGT1A4 enzyme (Corning, Inc. Life Science, Tewksbury, MA, USA) as an enzyme source and the UGT-Glo™ Screening System (Promega) according to the manufacturer’s protocol. The assay systems contain two glucuronidation reaction sets in parallel. An enzyme source of UGTs (UGT1A4, UGT1A1 or UGT2B7) and the proluciferin substrate (UGT1A4 specific substrate or UGT multienzyme substrate) were included both reactions. However, the uridine 5-diphosphoglucuronic acid (UDPGA) cofactor to generate the glucuronidated proluciferin was applied only in one reaction (plus-UDPGA reaction). In brief, 10 μL of 4-fold concentrated test samples, the positive controls (diclofenac (Sigma Chemical Co.) as an inhibitor of the UGT1A1 and UGT2B7 isozymes; lopinavir (Sigma Chemical Co.) as an UGT1A4 inhibitor) or vehicle (1% DMSO) were added to each well of white opaque 96-well plates (Costar® Corning Inc., NY, USA). Sequentially, 10 μL of 16 mM UDPGA or distilled water was added to the desired wells for plus-UDPGA or minus-UDPGA reactions, respectively. The enzyme reactions were started by adding 20 μL of 2× UGT reaction mixture containing UGT-Glo™ buffer, UGT substrate and UGT microsomes in the relevant wells. In the minus-UGT control wells, 20 μL of 2× control reaction mixture containing control microsomes instead of UGT microsomes was added. The plate was incubated for 120, 90 and 180 min with shaking at 37°C, respectively, for UGT1A1, UGT2B7 and UGT1A1. After the enzyme reaction, 40 μL of Luciferin Detection Reagent containing D-cysteine was added to all wells. To stabilize the luminescence signal, the plate was placed for 20 min at room temperature, and the luminescence intensity (LI) was later measured using a SpectraMax® i3. The percent inhibition was calculated by the following equation: % inhibition = [1 – (S/CAVR)] × 100, where S is the percent of substrate consumed (%SC) of each test sample or the minus-UGT control wells, and CAVR is the average %SC of the minus-UGT control wells. The %SC was calculated by the following equation: %SC = ΔLI/mLIAVR × 100, where ΔLI is the difference of background corrected LI at the minus-UDPGA reaction and the plus-UDPGA reaction for each well, and mLIAVR is the average LI of the minus-UDPGA reaction for each condition. To calculate the background corrected LI, the average LI of the minus-UGT control plus-UDPGA wells was subtracted from the average LI of the minus-UDPGA reaction, and then this LI value was added to all the remaining LI of plus-UDPGA reactions. The inhibitory effects of test samples on the activities of UGTs were created with concentration-dependent inhibition response curves by logistic 3 or 4 parameter curve fit function of SigmaPlot program. The data were described as the mean ± standard error of the mean (SEM) (n = 2).
Results
1. Anti-inflammatory effects in LPS-stimulated RAW 264.7 cells
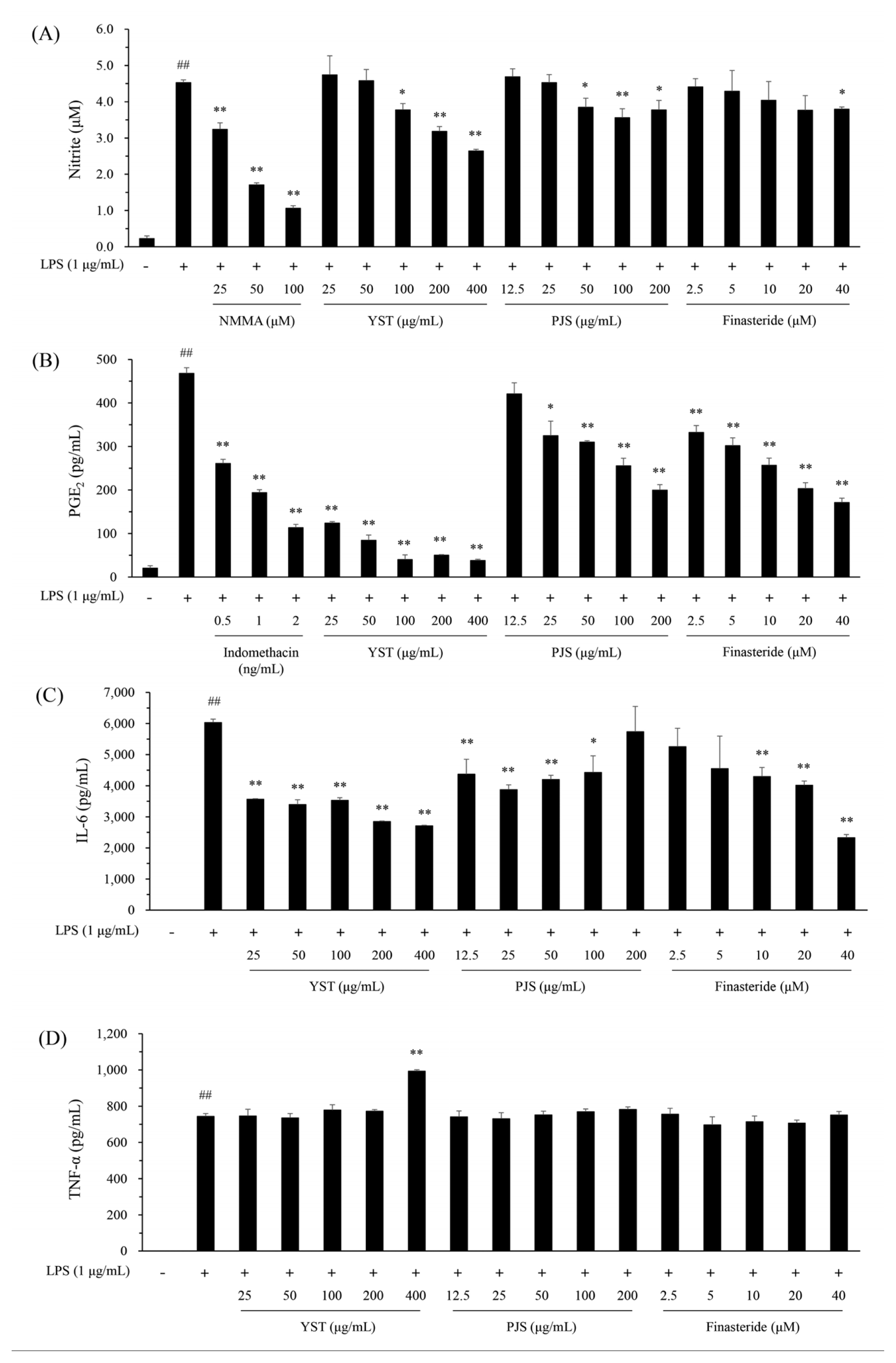
The inhibitory effects of YST, PJS and finasteride on the production of inflammatory mediators and cytokines in LPS-stimulated RAW 264.7 cells were investigated. YST inhibited the LPS-induced production of NO, PGE2 and IL-6 in RAW 264.7 cells in a dose-dependent manner (Fig. 1). The NO level was reduced to 56.25±1.02% of the level of LPS-treated cells by YST at a concentration of 400 μg/mL, which is the maximum concentration without cytotoxicity in RAW 264.7 cells (Fig. 1A and supplementary Fig. 1). PGE2 production was strongly inhibited by YST; the IC50 was below 25 μg/mL, which is the minimum concentration to treat the cells (Fig 1B). Concerning IL-6 production, the IC50 value of YST was 104.21 μg/mL (Fig. 1C). PJS inhibited NO and PGE2 production in a dose-dependent manner, and the IC50 value of PJS on PGE2 inhibition was 106.15 μg/mL (Fig. 1A and 1B). The maximum inhibitory percentage of NO production was 22.50±5.77% at 100 μg/mL PJS compared to the LPS-treated cells (Fig. 1A). IL-6 levels were reduced in a dose-dependent manner at low concentrations (12.5 and 25 μg/mL, p<0.01) of PJS; however, the level of IL-6 gradually increased at over 50 μg/mL and finally became similar to the level of LPS-treated cells at 200 μg/mL (Fig. 1C). Finasteride, a typical medication to treat BPH, inhibited PGE2 and IL-6 production in a dose-dependent manner. The IC50 values for PGE2 and IL-6 production were 11.54 and 30.90 μM finasteride (Fig. 1B and 1C). The NO level was significantly reduced at 40 μM finasteride (p<0.05), however, the percent inhibition was very low, 13.03±1.43% compared to the LPS-treated cells (Fig. 1A). There were no inhibitory effects on LPS-induced TNF-α production upon treatment of YST, PJS and finasteride in RAW 264.7 cells (Fig. 1D). The IC50 values of positive controls, NMMA and indomethacin, which inhibit LPS-stimulated NO and PGE2 production, were 35.28 μM and 0.617 ng/mL, respectively.
2. Inhibitory effects on the activities of human drug metabolizing enzymes (DMEs)
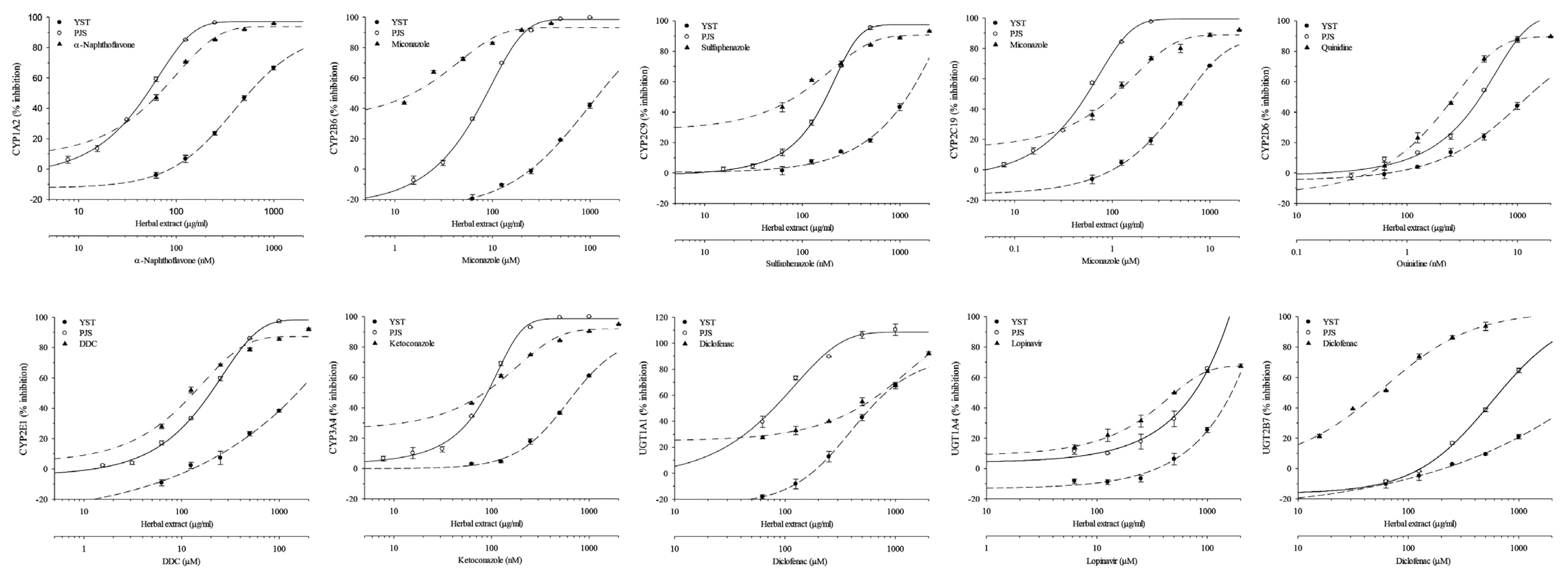
Change in enzyme activities caused by YST and PJS were measured on DMEs. YST inhibited the activities of 7 CYP450s and 3 UGTs in a dose-dependent manner (Fig. 2). The IC50 values of YST on UGT1A4 and UGT2B7 were 49.35 and 165.17 μg/mL, respectively, whereas other enzymes were slightly inhibited, since IC50 values were over than 300 μg/mL. The IC50 values of CYP1A2, CYP2C19 and CYP3A4 were 555.81, 601.62 and 720.74 μg/mL, respectively (Table 3). However, the IC50 values of CYPB6, CYP2C9, CYP2D6 and CYP2E1 were could not be calculated because they were more than 1000 μg/mL. The inhibitory effects of PJS were relatively strong compared to the effects of YST on DMEs. The 10 DMEs were inhibited by PJS in a dose-dependent manner (Fig. 2). There was relatively strong inhibition by PJS on CYP1A2 and CYP2C19 activities with IC50 values of 49.07 and 53.84 μg/mL, respectively. PJS was a moderate inhibitor of CYP2B6, CYP2C9, CYP2E1, CYP3A4 and UGT1A with IC50 values of 84.89, 170.54, 191.06, 85.86 and 77.03 μg/mL, respectively. PJS negligibly inhibited CYP2D6, UGT1A4 and UGT2B7 activities with their IC50 values being higher than 300 μg/mL (Table 3).
Discussion
The cause of BPH development in men has not been thoroughly elucidated; however, it is presumed that sex hormone changes caused by aging, such as increasing levels of DHT, which has higher affinity for the androgen receptor than testosterone; metabolic syndromes, including obesity and cardiovascular risk factors; and chronic prostatic inflammation20–22). Infiltration of inflammatory cells, including T and B lymphocytes, macrophages, and mast cells was in most TURP specimens from BPH patients was reported23,24). Management methods for BPH are classified as follows: 1) watchful waiting, 2) medical treatments, including mono and combination drug therapy, 3) surgical treatments such as TURP and simple prostatectomy, and 4) device therapies such as transurethral microwave thermotherapy (TUMT), transurethral needle ablation (TUNA), urethral stent, and prostatic urethral lift22). A portion of patients are not only unsatisfied with the therapeutic efficacies of conventional medications but also have concerns regarding adverse effects. Herbal medicines have a long history of being used to treat several diseases, and might be considered safer than synthesized chemical drugs3). Monotherapy of herbal remedies and combination therapies with conventional medications in patients with BPH is increasingly being used25). Therefore, the combination of finasteride, an inhibitor of 5α-reductase, with herbal formulas, which have anti-inflammatory effects, may be more effective to treat BPH. In the present study, anti-inflammatory activities of the herbal formulas YST and PJS and the drug finasteride were evaluated in LPS-stimulated RAW 264.7 cells. The production of NO, PGE2, and IL-6 were inhibited by treatment of YST, PJS, and finasteride. However, TNF-α was not inhibited by YST, PJS, and finasteride. Lim et al. have reported that YST inhibits NO and IL-6 production in LPS-stimulated RAW 264.7 cells, even though the composition of YST was slightly different from that in the present study26).
Williams et al., reported that, as of 2002, approximately 75% of the top 200 drugs prescribed in the United States are eliminated by DMEs, that approximately 70% of these drugs are metabolized by the CYP450s (phase I DMEs), that approximately 15% of these drugs are metabolized by the UGTs (phase II DMEs)27). Treating chronic diseases with multiple drugs has increased because most chronic diseases develop with several complications. However, the use of multiple drugs increases the risk of side effects such as reducing the efficacy and increasing the toxicity of drugs by drug-drug interactions, herb-drug interactions, and drug-food interactions28). Guidelines for drug-drug interaction (DDI) research are presented in each country, such as the US food and drug administration (FDA) and the European medicines agency (EMA) guidelines29), but there is still no standard method for herb-drug interaction (HDI) research, and HDI-related studies are lacked compared to DDI. Assessing the influence of herbal medicines on the activity of DMEs in an in vitro system is a basic tool to predict a potential of HDI30).
In the present study, the influence of the herbal formulas YST and PJS, which have been used to treat BPH, on activities of seven CYP450s and three UGTs from human microsomes were assayed to predict herbal-drug interactions. YST inhibited the activities of all enzymes, however the IC50 values for the enzymes, except for UGT1A4, were higher than 100 μg/mL, a concentration that might not influence the metabolism of other drugs. The activity of UGT1A4 was inhibited with an IC50 value of 49.35 μg/mL by YST. PJS inhibited the activities of CYP1A2, CYP2B6, CYP2C19, CYP3A4, and UGT1A1 with IC50 values below 100 μg/mL. Finasteride is excreted via two metabolic pathways mediated by as follows; 1) CYP3A4 and UGT1A4, and 2) CYP3A4 and UGT1A3. Finasteride is converted to ω-hydroxyfinasteride and hydroxyfinasteride glucuronide through the first pathway, and converted to finasteride-ω-oic acid and finasteride carboxylic acid glucuronide via the other pathway, respectively31). Finasteride does not appear to influence CYP450-mediated drug metabolism. Although PJS and YST inhibited the activities of CYP3A4 and UGT1A4, respectively, they might not influence the metabolism of finasteride because the IC50 values of herbal formulas on drug metabolizing enzymes are too high to affect the metabolism. Our results suggest that the combination with finasteride and YST or PJS might not influence drug metabolism of each other and we expected there to be synergistic effects to treat BPH.