Management of Idiopathic Interstitial Pneumonia Treated only with Traditional Korean Medicine: a Case Report
Article information
Abstract
Objectives:
To present the case of a 76-year-old man diagnosed with idiopathic interstitial pneumonias (IIPs) treated exclusively with traditional Korean medicine (TKM).
Methods:
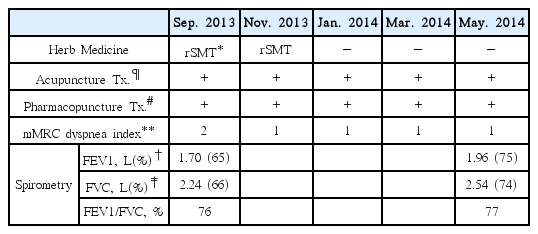
The patient was treated with the revised Sasammaekmundong-tang (rSMT), acupuncture, and pharmacopuncture from September 2013 to May 2014 in our outpatient clinic. We prescribed rSMT three times a day for 4 months. Acupuncture and pharmacopuncture were performed twice a week for 9 months. To evaluate therapeutic efficacy, we recorded the patient’s chief complaints at each visit, and pulmonary function test was performed at intervals of three months. Chest radiography and chest computed tomography were performed before and after treatment, respectively.
Results:
During the course of the treatment, dyspnea, cough, sputum, and overall pulmonary function improved.
Conclusions:
We suggest that traditional Korean medicine (TKM) for IIPs patients might be effective in the control of the main symptoms of IIPs and in the recovery of respiratory function.
Introduction
Idiopathic interstitial pneumonias (IIPs) are a heterogeneous group of diffuse parenchymal lung diseases, also known as interstitial lung diseases.1) IIPs can be classified as chronic fibrosing interstitial pneumonia (IP), smoking-related IP, and acute/ subacute IP. Chronic fibrosing IP includes idiopathic pulmonary fibrosis (IPF) and idiopathic nonspecific interstitial pneumonia (NSIP).2) So far, there is no specific treatment for IPF, which has shorter survival than other IIPs.3) The prognosis of idiopathic NSIP varies from individual to individual; some evolve to end-stage fibrosis and even death. Differentiation of NSIP from IPF is very important because the two subtypes have different treatment and prognosis. Histologically, IPF is characterized by the presence of honeycombing and subpleural distribution; NSIP is characterized by peribronchovascular distribution, wide areas of ground-glass attenuation, and subpleural sparing.4–6)
In traditional Korean medicine (TKM), IIPs may be defined by a combination of diseases. These include dyspnea
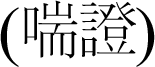 , dry cough
, dry cough
 , lung consumption
, lung consumption
 , and lung atrophy
, and lung atrophy
 . The pattern identifications on IIPs are as follows: lung qi deficiency syndrome
. The pattern identifications on IIPs are as follows: lung qi deficiency syndrome
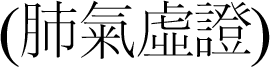 , syndrome of dual deficiency of qi and yin
, syndrome of dual deficiency of qi and yin
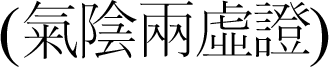 , syndrome of stagnant of qi-blood
, syndrome of stagnant of qi-blood
 , syndrome of dual deficiency of yang and yin
, syndrome of dual deficiency of yang and yin
 .7)
.7)
Several researchers have investigated the benefits of herbal medicines for interstitial lung disease in Korea.8–14) In addition, Son et al. reported on a patient with IIPs who was treated with Taeumjowi-tang and Bopae-tang.15)
However, there are not many clinical reports on IIPs with TKM so far.
Here, we present the case of a 76-year-old man diagnosed with IIPs who showed improved respiratory function after being treated exclusively with TKM.
Case Report
In September 2013, a 76-year-old man was admitted to our outpatient clinic because of dyspnea, cough, and sputum persisting for 3 months. He was previously diagnosed with IPF or NSIP by chest computed tomography (CT) at the local university hospital in July 2013. After the diagnosis, he was recommended further histological examination and steroid treatment. The patient refused treatment, but agreed to receiving TKM alone. He had been diagnosed with hypertension and diabetes mellitus (20 years prior), ischemic heart disease (4 years prior) and Parkinson’s disease (2 years prior), and had taken oral medications related to the above-mentioned diseases (irbesartan, nicorandil, isosorbide mononitrate, Aspirin, clopidogrel, rosuvastatin, sitagliptin/metformin and Stalevo). In addition, he was an ex-smoker (had smoked for 40 years). On the first visit to our outpatient clinic, the patient’s vital signs were as follows: blood pressure, 110/70 mmHg; pulse rate, 80 beats per minute; and respiratory rate, 20 breaths per minute. Chest auscultation revealed inspiratory crackles in both lower lung fields and no cardiac murmur was noted. Abdominal examination was unremarkable. The patient’s laboratory test results were as follows: hemoglobin, 13.5 g/dL; white blood cell count, 5,200 cells/μL; platelet count, 178,000 cells/μL; and erythrocyte sedimentation rate, 13 mm/h; all other blood parameters were within normal range. Chest X-ray (PA) and chest CT showed multifocal fibrosis, honeycomb, and air-space consolidation with ground-glass opacity (GGO) bilaterally in subpleural regions and the lower pulmonary lobes (Fig. 1A and 1B).

Chest X–ray (PA) and chest CT in the case
Fig. 1A, 1B: Initial Chest CT scan and chest X–ray (PA) showed multifocal fibrosis, honeycomb and air–space consolidation with ground glass opacity (GGO) at bilateral subpleural and lower lungs. (July, 2013)
Fig. 1C, 1D: Chest CT scan and chest X–ray (PA) obtained at 8 months follow–up showed improvement of acute process of interstitial pneumonia. But comparing with July 2013 CT, there was no change in multifocal fibrosis and honeycomb at bilateral subpleural and lower lungs. (April, 2014)
Pulmonary function tests showed mild restrictive patterns with the following results: forced vital capacity (FVC), 66% of the predicted value (2.24 L); forced expiratory volume in 1 s (FEV1), 65% of the predicted value (1.70 L); and FEV1/FVC ratio, 0.73.
The tongue of the patient was red with dry fur and the pulse was fine/rapid. In the pattern identification, the patient was finally diagnosed as lung-stomach yin deficiency. Based on this diagnosis, we prescribed the revised Sasammaekmundong-tang (rSMT) three times a day to strengthen lung qi and enhance the respiratory function.
The composition of rSMT was as follows: 12 g each of Radix Codonopsis and Liriopis Tuber; 8 g each of Polygonati Odorati Rhizoma, Radix Stemonae and Morus Alba; 6 g each of Trichosanthes Kirilowii and Dolichoris Semen; 4 g each of Radix Glycyrrhizae, Radix Ginseng and Farfarae Flos; and 2 g each of Radix Asteris Radix and Fructus Schizandrae.
The patient also underwent acupuncture and pharmacopuncture treatment twice per week for 4 months, and then once a week for another 5 months. The main acupoints were selected as per TKM theory: LI4
 , LU10
, LU10
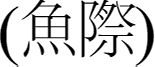 , LU7
, LU7
 , K3
, K3
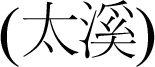 and SP6
and SP6
 . We used disposable stainless-steel needles (0.25 × 30 mm; Dong Bang Acupuncture Co., Korea). Each acupuncture session took 20 min. The type of pharmacopuncture was the hominis placenta pharmacopuncture (HPP; self-prepared in our clinic); 0.1 cc was injected per acupoint for a total of 0.5 cc. Acupoints for pharmacopuncture treatment were CV22
. We used disposable stainless-steel needles (0.25 × 30 mm; Dong Bang Acupuncture Co., Korea). Each acupuncture session took 20 min. The type of pharmacopuncture was the hominis placenta pharmacopuncture (HPP; self-prepared in our clinic); 0.1 cc was injected per acupoint for a total of 0.5 cc. Acupoints for pharmacopuncture treatment were CV22
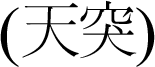 , BL13
, BL13
 and
and
 (extra point, 0.5 cm lateral to GV14).
(extra point, 0.5 cm lateral to GV14).
Two weeks after initiation of treatment, symptoms such as dyspnea and sputum gradually resolved. Therefore, the herbal medicine prescription was maintained. Two months later, dyspnea had improved notably. Symptoms such as cough and sputum disappeared four months after administration of rSMT. Hence, we discontinued the herbal medicine, and reduced the acupuncture and pharmacopuncture treatment to once a week.
On April 19, 2014, chest radiograph showed an improvement of the acute process of interstitial pneumonia. However, compared with the results of chest CT of the previous year, there was no change in multifocal fibrosis and honeycomb in bilateral subpleural regions and the lower pulmonary lobes (Fig. 1C and ID).
After 9 months of treatment, FEV1 and FEV2 increased by 10% and 8%, respectively. The peak expiratory flow (PEF) was increased from 300 L/min to 380 L/min (Table 1 and Fig. 2).
Discussion
According to the 2008 Korean national survey of IIPs, the most frequent subtype was IPF (77.1%), followed by NSIP (11.9%). The 3-year survival rate was 62% for IPF patients; the 5-year survival rate was 85% for NSIP patients.16)
IPF most commonly appears between the fifth and seventh decades of life, with two-thirds of all cases arising in patients over 60 years of age.2) The mean age at presentation is 66 years. IPF occurs infrequently in those younger than 40 and rarely affects children, if at all.17) A typical chest CT of IPF is characterized by patchy, predominantly peripheral, predominantly subpleural, bibasilar reticular opacification. In contrast, NSIP is characterized by homogeneously thickened interstitial spaces that contain accumulated fibrosis and inflammation.18) NSIP occurs mainly in middle-aged adults. The average age at onset is 49 years, although individuals at both extremes of life can be affected. Unlike IPF, NSIP may occur in children. In Katzenstein and Fiorelli’s study,5) 45% of patients recovered completely, and 42% remained stable or improved; the median survival was greater than 13 years. The prognosis of NSIP is variable but generally good, with a positive response to steroids in most patients compared with IPF. Compared with NSIP, IPF is typically progressive and leads to significant disability. The median survival is 2 to 5 years from the time of diagnosis.19,20) Our patient could be diagnosed as IPF based on the radiological and epidemiological features of IPF.
Medical therapy is ineffective in the treatment of IPF. New molecular therapeutic targets have been identified and several clinical trials are being conducted. Meanwhile, pulmonary transplantation remains a viable option for patients with IPF.
Unlike Western medicine, there is no specific term or definition that refers to the current understanding of IIPs in TKM. However, the clinical features of idiopathic interstitial lung disease share similarities with dyspnea
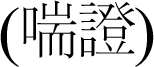 , dry cough
, dry cough
 lung consumption
lung consumption
 , and lung atrophy
, and lung atrophy
 .7)
.7)
Our patient was diagnosed with lung-stomach yin deficiency based on the TKM theory. Therefore, we decided to administer rSMT. Sasammaekmundong-tang has long been used to treat respiratory disease in Korea. It is recommended for patients who have a differential diagnosis of lung deficiency and lung qi and yin dual vacuity.
We added some herbs such as Radix Ginseng, Fructus Schizandrae, Radix Stemonae, Radix Asteris and Farfarae Flosin to the original formula.
In addition, acupuncture and pharmacopuncture were performed as complementary treatments. 9 months after treatment with TKM, the patient’s symptoms (cough, dyspnea and sputum) and respiratory function improved (Table 1).
However, conclusions should be drawn within the limitations of this single-case report. The diagnosis of IPF was not confirmed because lung biopsy was not performed. It is possible that this was actually a case of NSIP, which is known for having a good prognosis. Nevertheless, the improvement of respiratory function after such short TKM treatment is remarkable.
In conclusion, this case gives us some evidence that TKM offers beneficial effects (from an approach aimed at relieving symptoms) for patients with IIPs and Sasammaekmundong-tang used mainly in this case is as an effective regimen for IIPs.
Further studies are required to demonstrate the efficacy of TKM on interstitial lung diseases.
Acknowledgements
This study was supported by research funds of Daejeon University.