Skin tissue homogenate analysis for ceramide and TGF-β1 contents with TGF-β1 mRNA expressions after treatment of pomegranate concentrated solution and dried pomegranate concentrate powder in mice
Article information
Abstract
Objectives
The aim of this study was to solve skin moisturizing action mechanism issues of pomegranate concentrated solution (PCS) and dried pomegranate concentrate powder (PCP), at least partially.
Materials and methods
In this study, ceramide and TGF-β1 contents with TGF-β1 mRNA expressions were analysis on the skin tissue homogenate samples after 56 days of continuous oral administration of PCS 1, 2, and 4 ml/kg, and PCP 100, 200 and 400 mg/kg.
Results
Noticeable and dose-dependent increases of skin TGF-β1 contents and mRNA expressions were demonstrated in all PCP and PCS treated mice as compared with intact vehicle control, but no significant changes on the skin ceramide contents were demonstrated in all PCP and PCS treated mice as compared with intact vehicle control, in the current study. In addition, PCP 200 mg/kg showed similar increases of the skin TGF-β1 contents and mRNA expressions as compared to those of PCS 4 ml/kg.
Conclusions
The presented results suggested that in vivo skin moisturizing effects of PCP and PCS after oral administration through up regulation of hyaluronan synthesis demonstrated in our previous results, may be possibly mediated by modulation of TGF-β1 expressions at least partially, without critical influences on the skin ceramide contents.
Introduction
Hyaluronan is a high molecular weight linear glycosaminoglycan of alternating glucuronic acid and Nacetylglucosamine residues and is found in the extracellular matrix, especially of connective tissues such as dermis1). Hyaluronan has several physiochemical and biologic functions, such as space filling, lubrication, cell proliferation, and migration2). Skin contains about half of the total body hyaluronan3). Histological analysis showed that hyaluronan distributes to not only dermis but also epidermis4). Furthermore, the ability of keratinocytes to synthesize hyaluronan has been reported in both cell cultures5,6) and organ cultures7,8).
Pomegranate is a rich source of crude fibers, pectin, sugars, and several tannins9). In addition, it has been reported that pomegranate contains some species of flavonoids and anthocyanidins in their seed oil and juice, and shows an antioxidant activity three times more potently than red wine and green tea extract10,11). Furthermore, the chemopreventive and adjuvant therapeutic applications of pomegranate to human breast cancer have been warranted recently12). Owing to these significant biological activities, pomegranate juice is being increasingly popularized in world wide. Especially, we already reported that favorable in vivo and in vitro skin protective effects of pomegranate concentrated solution (PCS) including possible in vitro skin moisturizing effects13,14). In addition, PCS and dried pomegranate concentrate powder (PCP) increased the skin hyaluronan contents through up regulated hyaluronan synthase (Has) 1, 2 and 3, and showed related skin moisturizing effects15). However, it also pointed out that more detail mechanism studies and in vivo evidences for skin moisturizing effects of PCP and PCS should be further evaluated.
Hyaluronan is synthesized at the inner surface of the plasma membranes as long chains by adding sugar residues at the reducing end, and then is extruded to the extracellular space16,17). The hyaluronan synthase activity has been found to localize exclusively in plasma-membrane fractions of cultured human skin fibroblasts18,19). The synthesis of hyaluronan is stimulated by growth regulatory factors, such as TGF-β1, platelet-derived growth factor-BB, fibroblast growth factor, and epidermal growth factor20–22). Furthermore, it also reported that TGF-β up regulated the Has 1 and 21). In addition, ceramides play a key role in maintaining the structural integrity of the epidermal barrier and epidermal hydration23,24). In this study, therefore, ceramide and TGF-β1 contents with TGF-β1 mRNA expressions were analysis on the skin tissue homogenate samples, collected at our previous study15) to solve skin moisturizing action mechanism issues of PCS and PCP, at least partially.
Materials and methods
1. Test materials and animal experiments
Animals, test materials - pomegranate concentrated solution (PCS) and dried pomegranate concentrate powder (PCP), and tissue homogenate were used as same as our previous study15). Briefly, total 77 healthy male ICR mice (6-week old upon receipt) were prepared, and ten mice in each group were selected based on the body weight deviations after 7 days of acclimatization. Each of three different dosages of PCP and PCS were orally administered, once a day for 56 days, respectively. PCP was dissolved as 40, 20 and 10 mg/ml in distilled water, and orally administered in a volume of 10 ml/kg, equivalence to 400, 200 and 100 mg/kg of body weights, respectively. PCS was diluted by distilled water as 4:6, 2:8 and 1:9 (v/v) and orally administered in a volume of 10 ml/kg as equivalence to 4, 2 and 1 ml/kg, respectively. Some parts of the hair clipped dorsal skin tissues were separated, individually at 24 hrs after end of last 56th administration of test substances, and then tissue homogenates were prepared using bead beater (Model TacoTMPre, GeneResearch Biotechnology Corp., Taichung, Taiwan), ultrasonic cell disruptor (Model KS-750, Madell Technology Corp., Ontario, CA, USA) and radioimmunoprecipitation assay (RIPA) buffer (Sigma-Aldrich, St. Louise, MO, USA). After that the supernatants were separated by centrifugation at 15,000 rpm under 4 °C for 10 min. Supernatants were stored under −150 ºC conditions (Ultradeep freezer: MDF-1156, Sanyo, Tokyo, Japan) until analysis. All laboratory animals were treated according to the national regulations of the usage and welfare of laboratory animals, and approved by the Institutional Animal Care and Use Committee in Daegu Haany University (Gyeongsan, Gyeongbuk, Korea) prior to animal experiment [Approval No. DHU2015-067].
2. Ceramide content measurement
Ceramide concentrations on the skin tissue homogenates were determined using a Quick Start™ Bradford Protein Assay Kit (BioRad, Hercules, CA, USA) and primary antibody coated plate excluded blank ELISA kit (MyBioSource, San Diego, CA, USA). A 96-well plate was coated with 10 μg/well homogenates and incubated overnight at 4 °C. After washing, the wells were blocked with 200 μl/well assay diluent, and incubated with monoclonal anti-ceramide antibody (Sigma-Aldrich, St. Louise, MO, USA; dilution 1:10) for 2 hrs at room temperature. The wells were aspirated, washed three times, and incubated with horseradish-peroxidase-conjugated secondary antibody (100 μl) for 1 hr at 37 °C. 3,3′,5,5′-Tetramethylbenzidine substrate (90 μl) was added to each well and incubated for 30 min at 37 °C. Stop solution (50 μl) was added to each well, and the absorbance at 450 nm was measured using a microplate reader (Model Sunrise, Tecan, Männedorf, Switzerland), and skin tissue homogenate levels were calculated from standard ceramide (Sigma-Aldrich, St. Louise, MO, USA) calibration curves as μg/mg protein, according to previous established method24) with some modifications.
3. TGF-β1 content measurement
Skin tissue homogenate concentrations of TGF-β1 were measured using a commercial mouse TGF-β 1 ELISA kit (MyBioSource, San Diego, CA, USA), as pg/mg protein, according to the manufacturer’s instructions. Briefly, standards and samples (100 μl) were added to the appropriate wells and incubated for 2 hrs at 37 °C. The wells were aspirated and biotin-labeled antibody (100 μl) was added to each well and incubated for 1 hr at 37 °C. The wells were aspirated, washed three times, and incubated with horseradish peroxidase-conjugated avidin (100 μl) for 1 hr at 37 °C. 3,3′,5,5′-Tetramethylbenzidine substrate (90 μl) was added to each well and incubated for 30 min at 37 °C. Stop solution (50 μ l) was added to each well, and the absorbance at 450 nm was measured using a microplate reader25).
4. Realtime RT-PCR
Total RNA in the individual hair clipped dorsal back skins prepared at our previous study15), was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA), according to the method described in previous studies26,27). The RNA concentrations and quality were determined by CFX96TM Real-Time System (Bio-Rad, Hercules, CA, USA). To remove contaminating DNA, samples were treated with recombinant DNase I (Ambion, Austin, TX, USA). RNA was reverse transcribed using the reagent High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s instructions. Briefly, the cDNA strand first was synthesized from the total RNA and then the mixture of the primers and the cDNA products was amplified by PCR, and the conditions of PCR amplification were 58 °C for 30 min, 94 °C for 2 min, 35 cycles of 94 °C for 15 sec, 60 °C for 30 sec, 68 °C for 1 min, and then 72 °C for 5 min. Analysis was carried out using CFX96TM Real-Time System (Bio-Rad, Hercules, CA, USA), and their expression levels were calculated as relative to vehicle control. The expression of β-actin mRNA was used as a control for tissue integrity in all samples. The sequences of the PCR oligonucleotide primers were as listed in Table 1.
5. Statistical analysis
All values are expressed as mean ± SD of ten mice in this experiment. Multiple comparison tests for different dose groups were conducted. Variance homogeneity was examined using the Levene test. If the Levene test indicated no significant deviations from variance homogeneity, the obtain data were analyzed by one way ANOVA test followed by least-significant differences (LSD) multi-comparison test to determine which pairs of group comparison were significantly different. In case of significant deviations from variance homogeneity were observed at Levene test, a non-parametric comparison test, Kruskal-Wallis H test was conducted. When a significant difference is observed in the Kruskal-Wallis H test, the Mann-Whitney U (MW) test with Bonferroni’s correction was conducted to determine the specific pairs of group comparison, which are significantly different. Statistical analyses were conducted using SPSS for Windows (Release 14.0K, SPSS Inc., Chicago, IL, USA). Differences were considered significant at P < 0.05. In addition, the percent changes between intact vehicle control and test substance administered mice were calculated to help the understanding of the effects of PCP or PCS as [((Data of test material treated mice − Data of intact vehicle control mice)/Data of intact vehicle control mice) × 100], according to our previous report28).
Results
1. Skin ceramide content changes
No significant changes on the skin ceramide contents were observed in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mice as compared to those of intact vehicle control mice, at 24 hrs after end of last 56th administration (Fig. 1). The skin ceramide contents at 24 hrs after end of 56 days of continuous treatment in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mice were changed as −1.39, 3.69, −2.33, −2.07, 2.49 and 0.58% as compared with intact vehicle control mice, respectively.
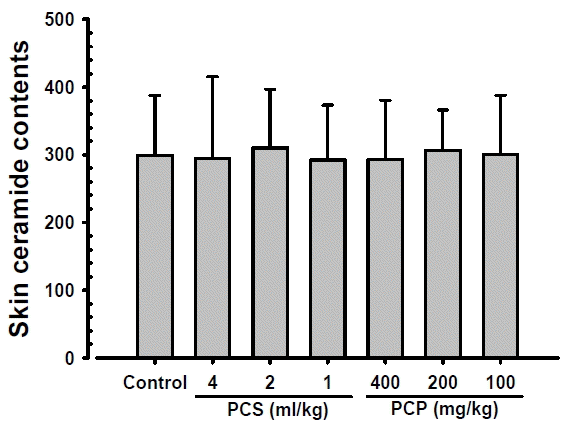
Skin Ceramide Contents. No significant changes on the skin ceramide contents were observed in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mice as compared to those of intact vehicle control mice, at 24 hrs after end of last 56th administration, in our experiment. Values are expressed mean ± S.D. of 10 mice, μg/mg protein. PCP = Dried pomegranate concentrate powder, PCS = Pomegranate concentrated solution.
2. Skin TGF-β1 content changes
Significant and dose-dependent increases of skin TGF-β1 contents were demonstrated in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mouse dorsal back skins as compared with intact vehicle control mice, at 24 hrs after end of last 56th administration (Fig. 2). The skin TGF-β1 contents at 24 hrs after end of 56 days of continuous treatment in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mice were changed as 35.98, 24.45, 19.85, 40.49, 35.42 and 23.16% as compared with intact vehicle control mice, respectively.
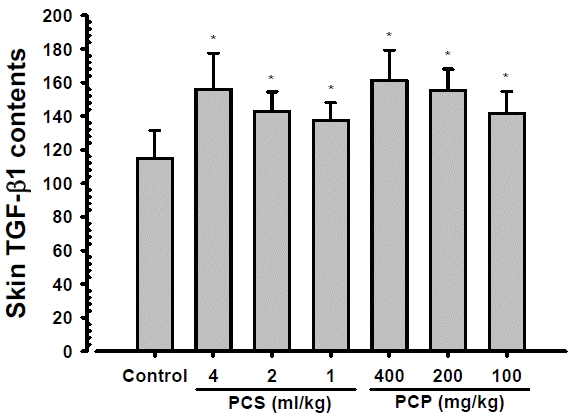
Skin TGF-β1 Contents. Significant and dose-dependent increases of skin TGF-β1 contents were demonstrated in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mouse dorsal back skins as compared with intact vehicle control mice, at 24 hrs after end of last 56th administration, in the current inspections. Values are expressed mean ± S.D. of 10 mice, pg/mg protein. PCP = Dried pomegranate concentrate powder, PCS = Pomegranate concentrated solution, TGF = Transforming growth factor. * p<0.01 as compared with intact vehicle control.
3. Changes on the skin TGF-β1 mRNA expressions
Significant (p<0.01) and dose-dependent increases of skin TGF-β1 mRNA expressions were demonstrated in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mouse hair clipped dorsal back skin tissue homogenates as compared with intact vehicle control mice, at 24 hrs after end of last 56th administration (Fig. 3). The skin TGF-β1 contents at 24 hrs after end of 56 days of continuous treatment in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mice were changed as 46.19, 32.54, 18.89, 69.24, 47.28 and 35.81% as compared with intact vehicle control mice, respectively.
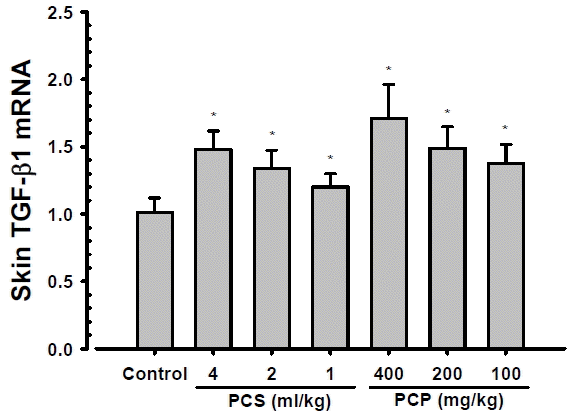
Skin TGF-β1 mRNA Expressions. Significant and dose-dependent increases of skin TGF-β1 mRNA expressions were demonstrated in PCS 4, 2 and 1 ml/kg, PCP 400, 200 and 100 mg/kg treated mouse hair clipped dorsal back skin tissue homogenates as compared with intact vehicle control mice, at 24 hrs after end of last 56th administration, in the current inspections. Values are expressed mean ± S.D. of 10 mice, relative expressions/β-actin mRNA. PCP = Dried pomegranate concentrate powder, PCS = Pomegranate concentrated solution, TGF = Transforming growth factor. * p<0.01 as compared with intact vehicle control by MW test.
Discussion
Various functional skin protective agents have been developed for anti-aging, whitening, wrinkle free, moisturizing and skin protective purposes in market29,30). Currently available functional skin protective agents for skin aging, however, have a number of limitations, such as economic problems, too expensive and exact or detail pharmacological and side effects are not fully understand15,31). Due to these factors, various efforts continuously attempt to search for affordable and effective functional ingredients with fewer side effects, especially on natural antioxidants13–15,30). In this study, ceramide and TGF-β1 contents with TGF-β1 mRNA expressions were analysis on the skin tissue homogenate samples after 56 days of continuous oral administration of PCS 1, 2, and 4 ml/kg, and PCP 100, 200 and 400 mg/kg, collected at our previous study15) to solve skin moisturizing action mechanism issues of PCS and PCP, at least partially. As results, noticeable and dose-dependent increases of skin TGF-β1 contents and mRNA expressions were demonstrated in all PCP and PCS treated mice as compared with intact vehicle control, but no significant changes on the skin ceramide contents were demonstrated in all PCP and PCS treated mice as compared with intact vehicle control, in the current study. In addition, PCP 200 mg/kg showed similar increases of the skin TGF-β1 contents and mRNA expressions as compared to those of PCS 4 ml/kg, at least in a condition of the present experiment.
As a bioactive lipid, ceramide has been implicated in a variety of physiological functions including apoptosis, cell growth arrest, differentiation, cell senescence, cell migration and adhesion32). Roles for ceramide and its downstream metabolites have also been suggested in a number of pathological states including cancer, neurodegeneration, diabetes, microbial pathogenesis, obesity, and inflammation33,34). Ceramides play a key role in maintaining the structural integrity of the epidermal barrier and epidermal hydration23,24). No significant changes on the skin ceramide contents were demonstrated in all PCP and PCS treated mice as compared with intact vehicle control, suggesting oral administration of PCP and PCS did not influenced on the skin ceramide contents, and skin moisturizing effects PCP and PCS oral administration, detected in our previous study15) may be mediated through hyaluronan rather than ceramide, at least in a condition of the current analysis.
Hyaluronan, a major extracellular matrix component of the skin with collagen, plays important roles in skin physiology including water homeostasis13,27,35), and synthesis by Has 1, Has 2 and Has 327,36,37). TGF-β plays a key role through regulating different cellular functions, and enhancing granulation tissue formation and collagen formation38). In addition, TGF-β has also been reported to be able to promote wound contraction through its direct induction of alpha smooth muscle act in expression in fibroblasts39,40). The synthesis of hyaluronan is stimulated by growth TGF-β120–22), and TGF-β up regulated the Has 1 and 21). In the present analysis, noticeable and dose-dependent increases of skin TGF-β1 contents and mRNA expressions were demonstrated in all PCP and PCS treated mice as compared with intact vehicle control, and PCP 200 mg/kg showed similar increases of the skin TGF-β1 contents and mRNA expressions as compared to those of PCS 4 ml/kg. These findings are considered as direct evidences that in vivo skin moisturizing effects of PCP and PCS after oral administration through up regulation of hyaluronan synthesis demonstrated in our previous results15), may be possibly mediated by modulation of TGF-β1 expressions at least partially, in a condition of the present skin tissue homogenate analysis
Notes
Conflicts of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
