References
3. Donini L. M., Busetto L., Bischoff S. C., Tommy C., Maria D. B., John A. B., Rocco B.. 2022;Definition and diagnostic criteria for sarcopenic obesity: ESPEN and EASO consensus statement. Obes Fact 15(3):321–335.
https://doi.org/10.1159/000521241.
4. Hong S. H., Choi K. M.. 2020;Sarcopenic Obesity, Insulin Resistance, and Their Implications in Cardiovascular and Metabolic Consequences. Int J Mol Sci 21(2):494.
https://doi.org/10.3390/ijms21020494.
5. Zhang X., Xie X., Dou Q., Liu C., Zhang W., Yang Y., Andy S. K.. 2019;Association of sarcopenic obesity with the risk of all-cause mortality among adult over a broad range of different settings: a updated meta-analysis. BMC Geriatr 19(1):183.
https://doi.org/10.1186/s12877-019-1195-y.
6. Chaoran L., Pui Y. W., Yik L. C., Simon K. C., Wing H. C., Sheung W. L., Ronald M. Y.. 2023;Deciphering the “obesity pardox” in the elderly: A systematic review and meta-analysis of sarcopenic obesity. Obes Rev 24(2):e13534.
https://doi.org/10.1111/obr.13534.
7. Goisser S., Kemmler W., Porzel S., Volkert D., Sieber C. C., Bollheimer L. C., Freiberger E.. (2015) Sarcopenic obesity and complex interventions with nutrition and exercise incommunity-dwelling older persons–a narrative review. Clin Interv Aging 10:1267.
https://doi.org/10.2147/CIA.S82454.
8. Poggiogalle E., Migliaccio S., Lenzi A., Donini L.. 2014;Treatment of body composition changes in obese and overweight older adults: insight into the phenotype of sarcopenic obesity. Endocrine 47(3):699–716.
https://doi.org/10.1007/s12020-014-0315-x.
9. Hit-contreras F. H., Bueno-Notivol J., Martínez-Amat A., Cruz-Díaz D., Hernadez A. V., Pérez-López F. R.. 2018;Effect of exercise alone or combined with dietary supplements on anthropometric and physical performance measures in community-dwelling elderly people with sarcopenic obesity: a meta-analysis of randomized controlled trials. Maturitas 116:24–35.
https://doi.org/10.1016/j.maturitas.2018.07.007.
10. Eglseer D., Traxler M., Schoufour J. D., Weijs P. J., Voortman T., Boirie Y., Bouer S., ; SO-NUT Consortium. 2023;Nutritional and exercise interventions in individuals with sarcopenic obesity around retirement age: a systematic review and meta-analysis. Nutr Rev 81(9):1077–1090.
https://doi.org/10.1093/nutrit/nuad007.
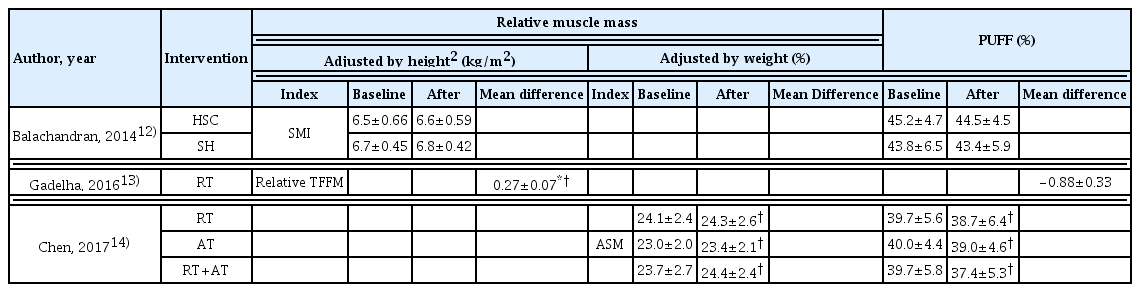
12. Balachandran A., Krawczyk S. N., Potiaumpai M., Signorile J. F.. 2014;High-speed circuit training vs hypertrophy training to improve physical function in sarcopenic obese adults: a randomized controlled trial. Exp Gerontol 60:64–71.
https://doi.org/10.1016/j.exger.2014.09.016.
13. Gadelha A. B., Paiva F. M., Gauche R., de Oliveira R. J., Lima R. M.. 2016;Effects of resistance training on sarcopenic obesity index in older women: a randomized controlled trial. Arch Gerontol Geriatr 65:168–173.
https://doi.org/10.1016/j.archger.2016.03.017.
14. Chen H. T., Chung Y. C., Chen Y. J., Ho S. Y., Wu H. J.. 2017;Effects of different types of exercise on body composition, muscle strength, and IGF-1 in the elderly with sarcopenic obesity. J. Am. Geriatr. Soc 65(4):827832.
https://doi.org/10.1111/jgs.14722.
15. Park J., Kwon Y., Park H.. 2017;Effects of 24-week aerobic and resistance training on carotid artery intima-media thickness and flow velocity in elderly women with sarcopenic obesity. J. Atheroscler. Thromb 24(11):1117–1124.
https://doi.org/10.5551/jat.39065.
16. Huang S. W., Ku J. W., Lin L. F., Liao C. D., Chou L. C., Liou T. H.. 2017;Body composition influenced by progressive elastic band resistance exercise of sarcopenic obesity elderly women: a pilot randomized controlled trial. European Journal of Physical and Rehabilitation Medicine 53(4):556–563.
https://doi.org/10.23736/S1973-9087.17.04443-4.
17. Liao C. D., Tsauo J. Y., Lin L. F., Huang S. W., Ku J. W., Chou L. C., Liou T. H.. 2017;Effects of elastic resistance exercise on body composition and physical capacity in older women with sarcopenic obesity: a CONSORT-compliant prospective randomized controlled trial. Medicine 96:23.
https://doi.org/10.1097/MD.0000000000007115.
18. Muscariello E., Nasti G., Siervo M., Di Maro M., Lapi D., D’Addio G., Colantuoni A.. 2016;Dietary protein intake in sarcopenic obese older women.(ORIGINAL RESEARCH). Clin. Interv. Aging 11:133.
https://doi.org/10.2147/CIA.S96017.
19. Sammarco R., Marra M., Di Guglielmo M., Naccarato M., Contaldo F., Poggiogalle E., Pasanisi F.. 2017;Evaluation of hypocaloric diet with protein supplementation in middle-aged sarcopenic obese women: a pilot study. Obes. Facts 10(3):160–167.
https://doi.org/10.1159/000468153.
20. Yin Y. H., Liu J. Y., Välimäki M.. 2023;Dietary behaviour change intervention for managing sarcopenic obesity among community-dwelling older people: a pilot randomised controlled trial. BMC Geriatrics 23:597.
https://doi.org/10.1186/s12877-023-04327-w.
21. Kemmler W., Teschler M., Weissenfels A., Bebenek M., Stengel S., Kohl M., Engelke K.. 2016;Whole-body electromyostimulation to fight sarcopenic obesity in community-dwelling older women at risk. Results of the randomized controlled FORMOsA-sarcopenic obesity study. Osteoporos. Int 27(11):3261–3270.
https://doi.org/10.1007/s00198-016-3662-z.
22. Kemmler W., Weissenfels A., Teschler M., Willert S., Bebenek M., Shojaa M., von Stengel S.. 2017;Whole-body electromyostimulation and protein supplementation favorably affect sarcopenic obesity in community-dwelling older men at risk: the randomized controlled FranSO study. (ORIGINAL RESEARCH) (Report). Clin Interv Aging 12:1503.
https://doi.org/10.2147/CIA.S137987.
23. Kim H., Kim M., Kojima N., Fujino K., Hosoi E., Kobayashi H., Yoshida H.. 2016;Exercise and nutritional supplementation on community-dwelling elderly Japanese women with sarcopenic obesity: a randomized controlled trial. J Am Med Dir Assoc 17(11):1011–1019.
https://doi.org/10.1016/j.jamda.2016.06.016.
24. Nabuco H. C., Tomeleri C. M., Fernandes R. R., Junior P. S., Cavalcante E. F., Cunha P. M., Barbosa D. S.. 2019;Effect of whey protein supplementation combined with resistance training on body composition, muscular strength, functional capacity, and plasma-metabolism biomarkers in older women with sarcopenic obesity: a randomized, double-blind, placebo-controlled trial. Clinical Nutrition ESPEN 32:88–95.
https://doi.org/10.1016/j.clnesp.2019.04.007.
25. Zhou X., Xing B., He G., Lyu X., Zeng Y.. 2018;The effects of electrical acupuncture and essential amino acid supplementation on sarcopenic obesity in male older adults: a randomized control study. Obes. Facts 11(4):327–334.
https://doi.org/10.1159/000491797.
26. Cruz-Jentoft A. J., Baeyens J. P., Bauer J. M., Boirie Y., Cederholm T., Landi F., Zamboni M., ; European Working Group on Sarcopenia in Older People. 2010;Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing 39:412–423.
https://doi.org/10.1093/ageing/afq034.
27. Newman A. B., Kupelian V., Visser M., Simonsick E., Goodpaster B., Nevitt M., Harris T. B., ; Health ABC Study Inverstigators. 2003;Sarcopenia: alternative definitions and associations with lower extremity function. Journal of the American Geriatrics Society 51:1602–1609.
https://doi.org/10.1046/j.1532-5415.2003.51534.x.
28. Chung J. Y., Kang H. T., Lee D. C., Lee H. R., Lee Y. J.. 2013;Body composition and its association with cardiometabolic risk factors in the elderly: A focus on sarcopenic obesity. Arch Gerontol Geriatr 56:270–278.
https://doi.org/10.1016/j.archger.2012.09.007.
29. Lim S., Kim J. H., Yonn J. W., kang S. M., Choi S. H., Park Y. J., Jang H. C.. 2010;Sarcopenic obesity: Prevalence and association with metabolic syndrome in the Korean longitudinal study on health and aging(klosha). Diabetes Care 33:1652–1654.
https://doi.org/10.2337/dc10-0107.
30. Janssen I., Heymsfield S. B., Ross R.. 2002;Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc 50:889–896.
https://doi.org/10.1046/j.1532-5415.2002.50216.x.
31. Chen L. K., Woo J., Assantachai P., Auyeung T. W., Chou M. Y., Iijima K., Arai H.. 2020;Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc 21:300–307.e2.
https://doi.org/10.1016/j.jamda.2019.12.012.
32. Studenski S. A., Peters K. W., Alley D. E., Cawthon P. M., McLean R. R., Harris T. B., Vassileva M. T.. 2014;The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci 69:547–558.
https://doi.org/10.1093/gerona/glu010.
33. Chen L. K., Liu L. K., Woo J., Assantachai P., Auyeung T. W., Bahyah K. S., Arai H.. 2014;Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc 15:95–101.
https://doi.org/10.1016/j.jamda.2013.11.025.
34. Ling C. H., de Craen A. J., Slagboom P. E., Gunn D. A., Stokkel M. P., Westendorp R. G., Maier A. B.. 2011;Accuracy of direct segmental multi-frequency bioimpedance analysis in the assessment of total body and segmental body composition in middle-aged adult population. Clin Nutr 30:610–615.
https://doi.org/10.1016/j.clnu.2011.04.001.
35. Ji Yeon Baek J. Y., Jung H. W., Kim K. M., Kim M., Park Y. C., Lee K. P., Lim J. Y.. 2023;Korean Working Group on Sarcopenia Guideline. Expert Consensus on Sarcopenia Screening and Diagnosis by the Korean Society of Sarcopenia, the Korean Society for Bone and Mineral Research, and the Korean Geriatrics Society. Ann Geriatr Med Res 27(1):9–21.
https://doi.org/10.4235/agmr.23.0009.
36. Kim K. K., Haam J. H., Kim B. T., Kim E. M., Park J. H., Rhee S. Y., Lee C. B., ; Committee of Clinical Parctice guidelines, Korean Society for the Study of Obestiy (KSSO). 2023;Evaluation and Treatment of Obesity and Its Comorbidities: 2022 Update of Clinical Practice Guidelines for Obesity by the Korean Society for the Study of Obesity. J Obes Metab Syndr 32(1):1–24.
https://doi.org/10.7570/jomes23016.
38. Garber C. E., Blissmer B., Deschenes M. R., Franklin B. A., Lamonte M. J., Lee I. M., Swain D. P., ; American College of Sports Medicine. 2011;American college of sports medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med Sci Sports Exerc 43:1334–1359.
https://doi.org/10.1249/MSS.0b013e318213fefb.
39. Schulte J. N., Yarasheski K. E.. 2001;Effects of resistance training on the rate of muscle protein synthesis in frail elderly people. Int J Sport Nutr Exerc Metab 11(Suppl):S111–118.
https://doi.org/10.1123/ijsnem.11.s1.s111.
41. Hunter G. R., Wetztein C. J., Mclafferty C. L. Jr, Zuckerman P. A., Landers K. A., Bamman M. M.. 2001;High-resistance versus variable-resistance training in older adults. Med Sci Sports Exerc 33:1759–1764.
https://doi.org/10.1097/00005768-200110000-00022.
43. Cho J. K., Kang H. S., Yoon J. H.. 2013;Increased Dietary intake of proteins for the prevention and Treatment of sarcopenic obesity in the Elderly. Korean J obes 22:77–82.
https://doi.org/10.7570/kjo.2013.22.2.77.
44. Morton R. W., Murphy K. T., McKellar S. R., Schoenfeld B. J., Henselmans M., Helms E., Phillips S. M.. 2017;A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br J Sports Med 52(6):376–384.
https://doi.org/10.1136/bjsports-2017-097608.
45. Sandberg M., Lundeberg T., Lindberg L. G., Gerdle B.. 2003;Effects of acupuncture on skin and muscle blood flow in healthy subjects. Eur J Appl Physiol 90:114–119.
https://doi.org/10.1007/s00421-003-0825-3.
46. Shinbara H., Okubo M., Kimura K., Mizunuma K., Sumiya E.. 2015;Contributions of nitric oxide and prostaglandins to the local increase in muscle blood flow following manual acupuncture in rats. Acupunct Med 33:65–71.
https://doi.org/10.1136/acupmed-2014-010634.
47. Zamboni M., Mazzali G., Zoico E., Harris T. B., Meigs J. B., Di Francesco V., Bosello O.. 2005;Health consequences of obesity in the elderly: a review of four unresolved questions. Int J Obes (Lond) 29(9):1011–1029.
https://doi.org/10.1038/sj.ijo.0803005.